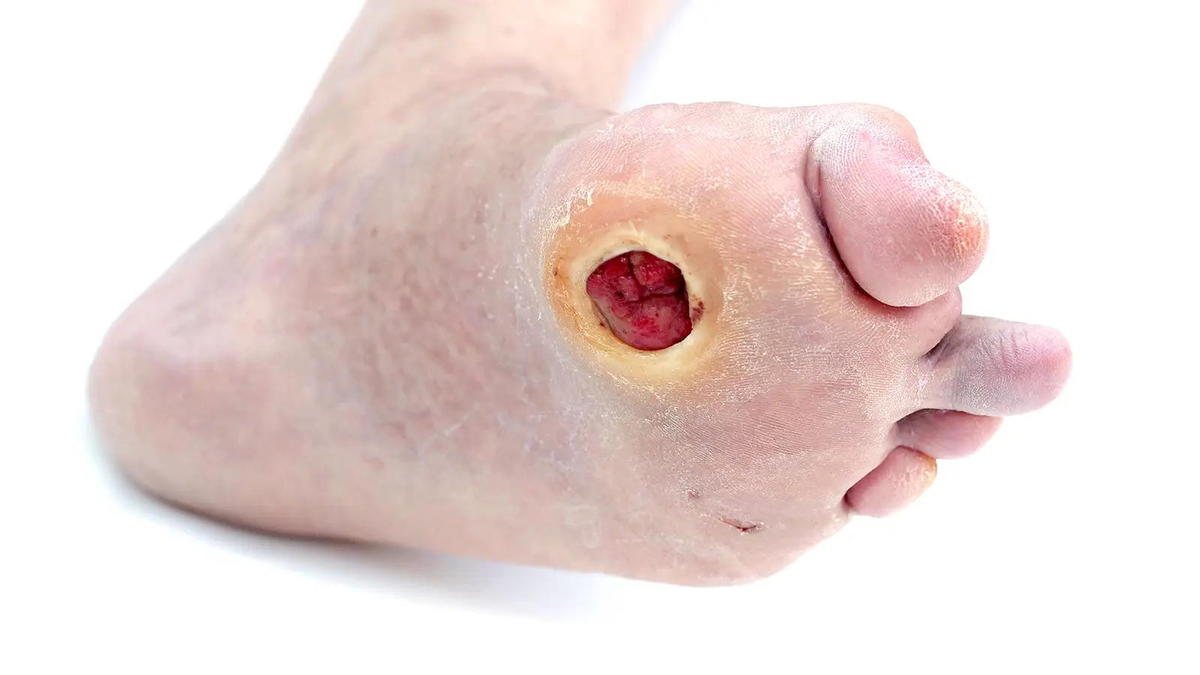
NOVEL DIABETIC FOOT ULCER CREAM SHOWS PROIMSE IN PHASE 3 TRIAL
posted: Sep. 14, 2021.

ON101 (Fespixon, Oneness Biotech), a first-in-class, macrophage-regulating, wound-healing cream for diabetic foot ulcers has shown benefit over absorbent dressings in a phase 3 trial, with another trial ongoing. The product became available in Taiwan on July 4, 2021, after receiving regulatory approval from the Taiwan Food and Drug Administration (FDA) based on efficacy and safety findings in a three-country phase 3 clinical trial.
 |
| Dr. Shawn Cazzell |
"Patients in the United States can access this new drug via the expanded access program or by participating in the second phase 3 trial in the United States," added co-author Shawn M. Cazzell, DPM, chief medical officer, Limb Preservation Platform, Fresno, California, who is involved with both trials. It is "exciting" to have a new therapy for diabetic foot ulcers, said Cazzell, because they are serious and life-threatening.
Source: Marlene Busko, Medscape News [9/10/21]
Courtesy of Barry Block, editor of PM News